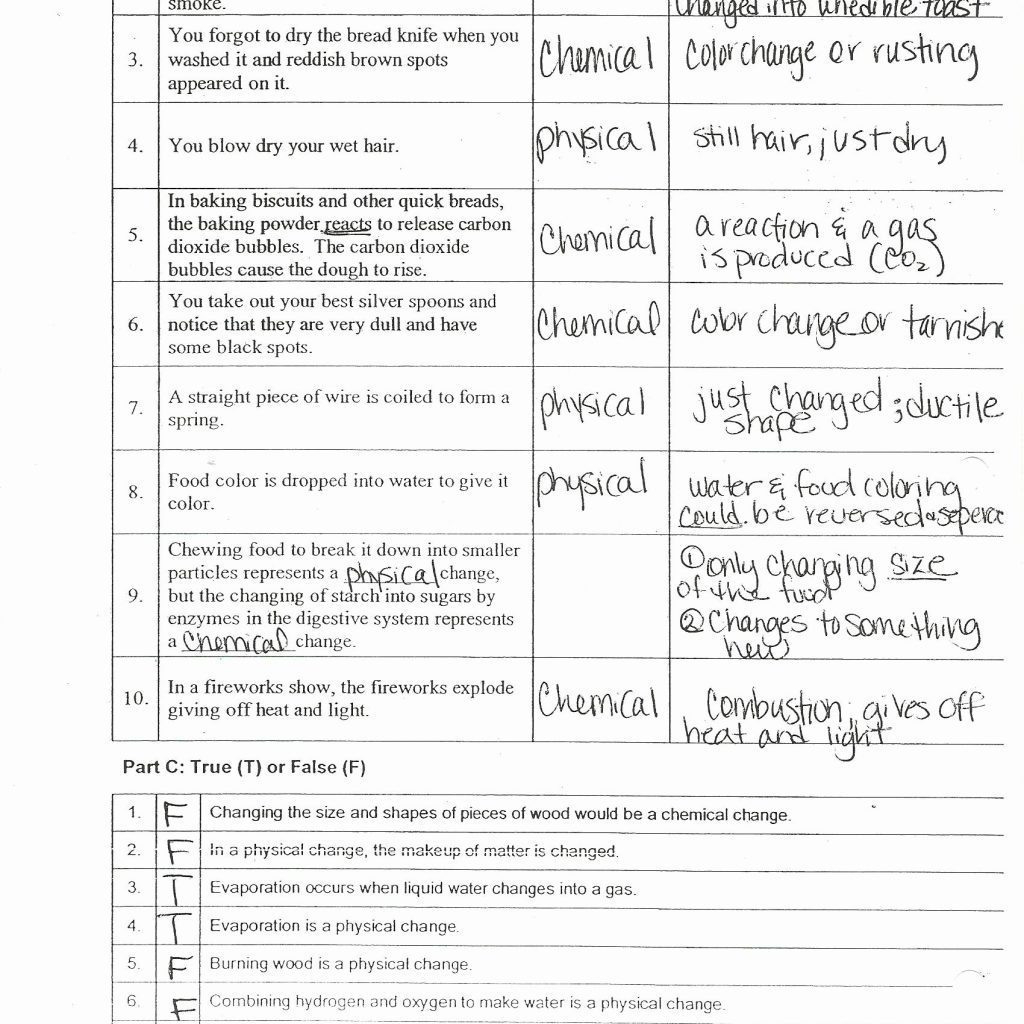
A lower pKa value indicates a stronger acid. pKa is the negative log of the acid dissociation constant or Ka value. The pKa value is one method used to indicate the strength of an acid. This allows the meter to recognize a particular pH buffer and calibrate with the correct buffer value at the measured temperature. pH meters contain pH versus temperature values for commonly used buffers. PH electrode measurements are made by comparing the readings in a sample with the readings in standards whose pH has been defined (buffers). When the pH is above 7, the solution is described as basic (or alkaline)-the activity of hydroxide ion is greater than that of hydrogen ion. When the pH is below 7, the solution is described as acidic-the hydrogen ion activity increases and the pH value decreases. A pH of 7 describes a neutral solution because the activities of hydrogen and hydroxide ions are equal. PH compares the acidity or alkalinity of a solution at a given temperature. Please see below for more detailed descriptions of the tests available at Triclinic Labs. Nitroglycerin is very dangerous because it explodes easily due to chemical reactivity neon poses almost no hazard because it is very unreactive. Iron, for example, combines with oxygen in the presence of water to form rust chromium does not oxidize. Examples of chemical properties include flammability, toxicity, acidity, reactivity (many types), and heat of combustion. The change of one type of matter into another type (or the inability to change) is a chemical property. In each of these examples, there is a change in the physical state, form, or properties of the substance, but no change in its chemical composition. We observe a physical change when wax melts, when sugar dissolves in coffee, and when steam condenses into liquid water Other examples of physical changes include magnetizing and demagnetizing metals (as is done with common anti-theft security tags) and grinding solids into powders (which can sometimes yield noticeable changes in color and consistency). A physical change is a change in the state or properties of matter without any accompanying change in its chemical composition. Other physical properties, such as the melting temperature of iron or the freezing temperature of water, can only be observed as matter undergoes a physical change. We can observe some physical properties, such as density and color, without changing the physical state of the matter observed. Familiar examples of physical properties include density, color, hardness, melting, and boiling points, and electrical conductivity. A physical property is a characteristic of matter that is not associated with a change in its chemical composition. The characteristics that enable us to distinguish one substance from another are called properties. Water content by Karl-Fischer and Dynamic Vapor Sorption.Ultraviolet and Visible Absorption Spectroscopy (UV/vis).Nuclear Magnetic Resonance Spectroscopy (NMR).Metals Analysis by Inductively Coupled Plasma Spectroscopy (ICP).High Performance Liquid Chromatography (HPLC, LC/MS and LC/MS/MS detection).Gas Chromatography - Mass Spectrometry (GC-MS).Elemental Microanalysis (Carbon, Oxygen, Hydrogen, Nitrogen, Sulphur / CHNS).These tests can be used to provide technical data such as identity, purity, impurity profile: 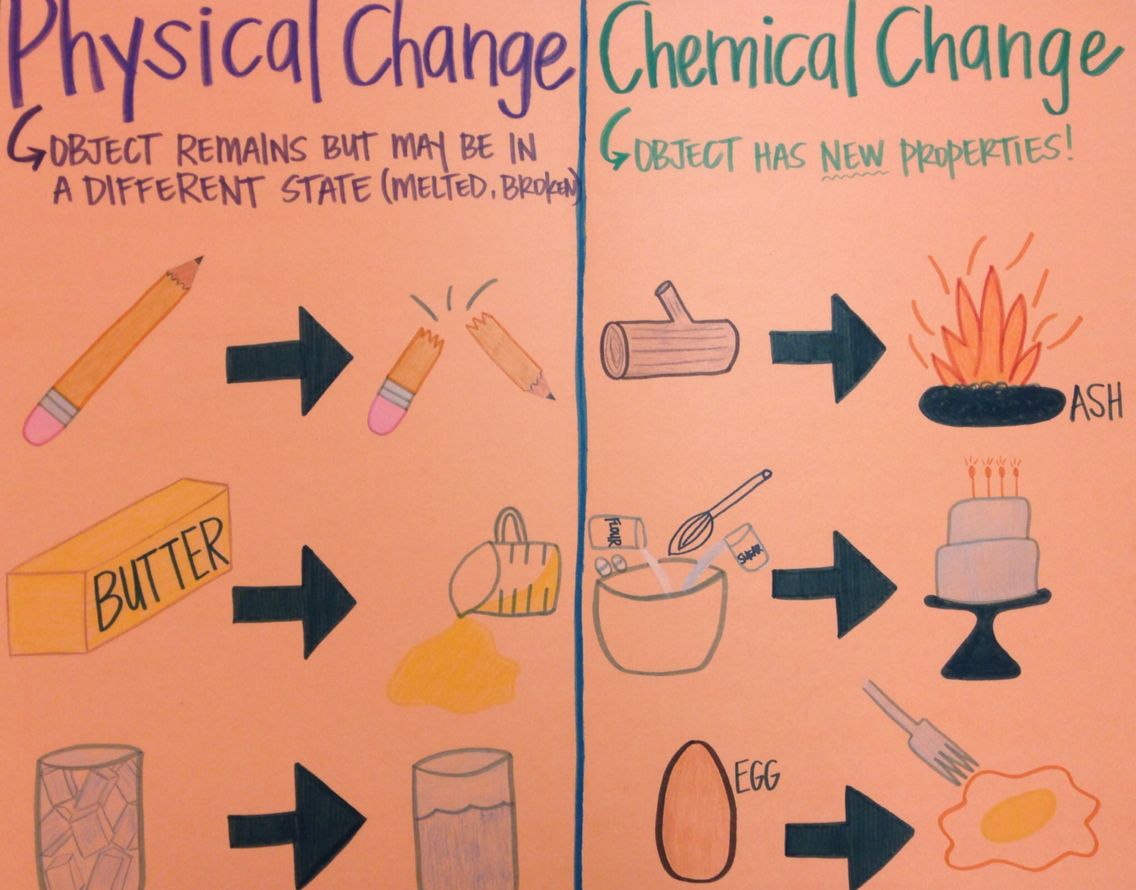
We offer the following services:Īdditional physico-chemical test measurements: Our lab is fully equipped to help you demonstrate control and consistency. Physical chemistry testing is carried out to attain regulatory compliance and can also be used for the purposes of research and development, product development, and technical support.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
